LIFE SCIENCES
Boost productivity & progress with compliant Life Science Services
Our Life Science Services create work environments that foster innovation and productivity while ensuring full regulatory compliance. We support your teams across diverse processes so they can focus on their core competencies and save valuable resources.
Why ISS for Life Sciences
Center of Excellence
Benefit from our Life Sciences CoE and extensive industry experience.
Tailored to your needs
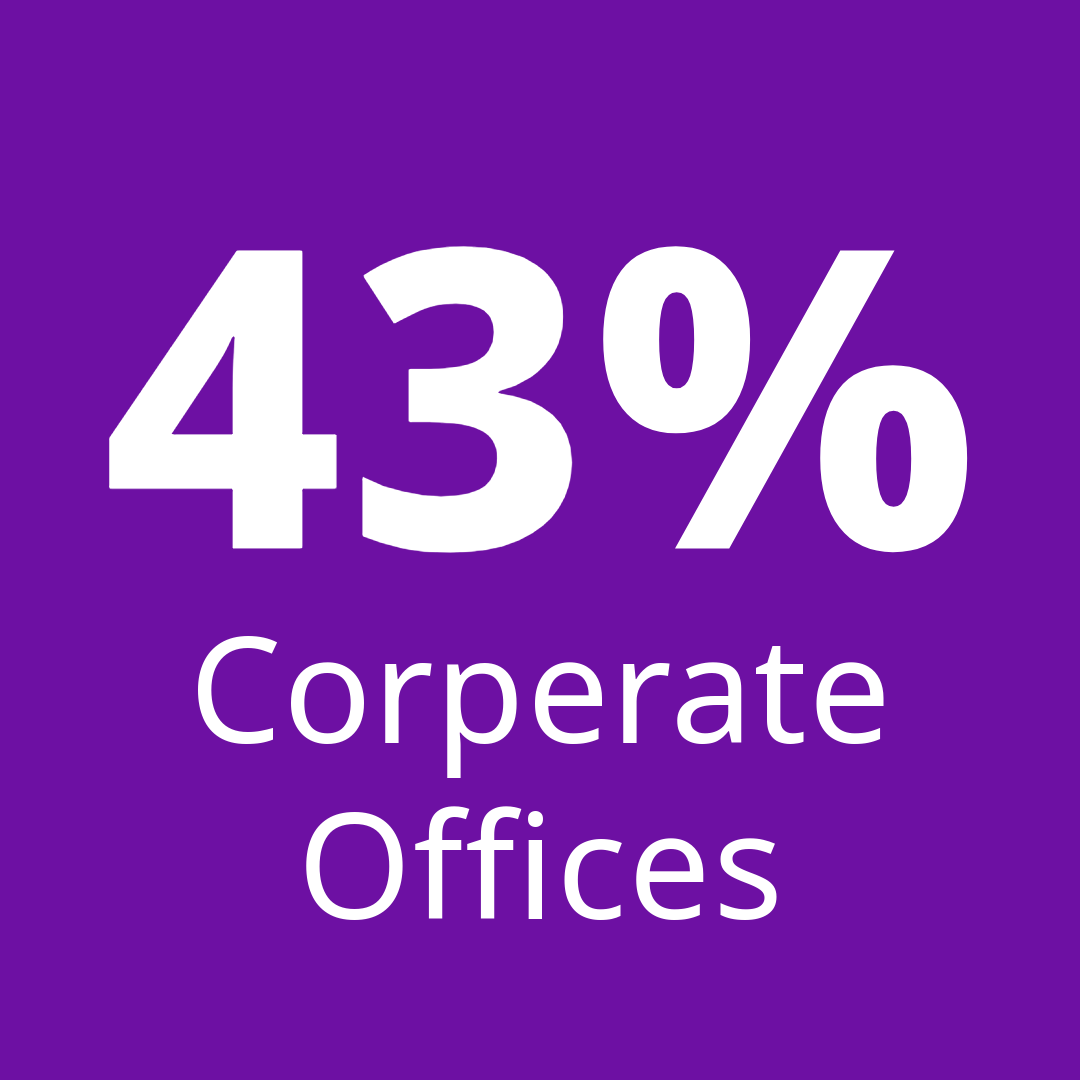
Customized service designs aligned to your processes and risk profile.
Comprehensive solutions from a single source, backed by deep industry know‑how and a proven compliance culture.
Compliance excellence
Internal expertise in Quality Control and HSE ensures GDP/GMP conformity.
Single-source delivery
Integrated life sciences solutions that reduce complexity and handovers.
What you get
Three core service lines that unlock quality, efficiency, and compliance.
GDP – certified Pharma Logistics
Leverage our certified GDP logistics service for secure, reliable pharmaceutical transportation throughout Europe. Strict temperature control and the highest hygiene standards come as standard.
Qualified drivers and validated routes
Continuous temperature monitoring
Traceability and documentation
Specialized GMP-compliant cleaning delivered by trained personnel, supported by a pharmaceutical‑grade quality management system and professional training program.
Auditable processes and SOPs
Cleaning in GMP zones A-D and ISO 9-5
Deviation handling & CAPA
Laboratory Support Services
Free up valuable time for your scientists. Our specialists handle defined lab support tasks so your team can focus on research, quality assurance, and control—while we ensure seamless local coordination.
Sample logistics & media prep
Inventory & consumables management
Equipment and Calibration Management
GMP – Services
„Through our Lab Services, your lab staff will experience immediate relief, freeing up valuable time to focus on advancing science.“
Articles & Insights
Thought leadership from ISS Switzerland’s Life Sciences team.
Smart Solutions for the Life Sciences Sector
ISS has proven itself over many years as a reliable FM service partner in the highly demanding life sciences environment. Today, the operational organization unit has 620 employees who deliver highly specialized solutions and ensure strict compliance with regulations.
How ISS enables cutting-edge Life Sciences research
ISS has proven itself over many years as a reliable FM service partner in the highly demanding life sciences environment. Today, the operational organization unit has 620 employees who deliver highly specialized solutions and ensure strict compliance with regulations.
How we can help
ISS has spent decades working with leading pharmaceutical companies at the forefront of science. We understand what it takes to design and maintain a work environment that fosters innovation, minimizes risk, achieves compliance, and attracts the boldest and brightest minds in the industry.
Get in contact
Tell us about your site, processes, and regulatory needs. Our life sciences team will respond within one business day.
-

Angela Behrend
Head Center of Excellence & Project Management
Dipl. Architect
20 years of experience in Facility Management with a strong background in architecture and planning.
Expertise in transition management, service development, and construction-accompanying FM.
References include Novartis, Biozentrum, Main Campus, and various life sciences mandates. -

Dr. Michael Eberhardt
Unit Lead Life Sciences Excellence
Ph.D. – Biomedical Research
15 years of experience in GMP and GxP environments, including the development of PQMS (Process Quality Management Systems).
Strong background in medical and biological research.
References include Novartis, Biozentrum, Main Campus, and various life sciences mandates. -

Martin Hainz
Lead HSE & Quality Management
Safety Engineer EKAS | Certified Quality Manager
Expert in occupational safety with dual qualifications as EKAS Safety Specialist and Quality Manager (DIN ISO 45001).
Extensive experience in health & safety, risk management, and quality systems in regulated environments.
References include Novartis, Biozentrum, Main Campus, Solvias, and various life sciences mandates. -

Beat Nickler
Lead GxP / QA Life Siences
Chemical Laboratory Technician
15 years of experience in pharma logistics, hazardous materials handling, lab renovations, and HSE in laboratory environments.
Strong operational background in regulated life sciences settings.
References include Solvias, Novartis, TF, and Main Campus.